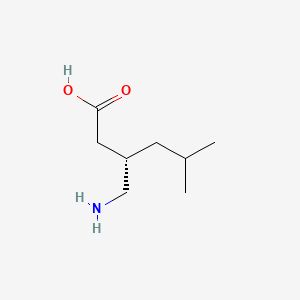
Bhiwandi, Thane, Maharashtra
- GST NO. : 27AKFPM8972N1Z9
View Mobile Number
| Business Type | Manufacturer, Exporter, Supplier, Retailer |
| Grade | IP / BP / USP / EP |
With characteristics like unmatched standard and superior grade, our range of product is the finest one obtainable in Mumbai(India). Our warehouse is installed with advanced machines and all required amenities ensuring safe storage of diverse kinds of products requiring different storage temperatures and conditions.
Details
-
Article - 1206-103
-
Batch No. - PG/023/16
-
Mfg. Date - 12/2016
-
Exp. Date - 11/2021
Remark : In the opinion of the undersigned the sample tested to above is of standard quantity of In House Specification and complies I.P. Specification with respect to the above mentioned test
Abstract From Factory Record
-
Analysed by - Q.C. Chemist
-
Approved by - Q . C. Manager
Disclaimer of liability : The information in this SDS was obtained from sources which we believe are reliable. However, the information is provided without any warranty, express or implied, regarding its correctness. The conditions or methods of handling, storage, use or disposal of the product are beyond our control and may be beyond our knowledge. For this and other reasons, we do not assume responsibility and expressly disclaim liability for loss, damage or expense arising out of or in anyway connected with the handling, storage, use or disposal of the product. Buyer should ascertain & verify the data and analyse on its own before using the product. This data I information is the indication and not final. Products covered by valid patents in a country are not offered & I or supplied there. The customer is responsible for verification of the patent position. Product are not offered in the countries where they are covered under Patent. However, ultimate responsibility remains of the customer.
Analytical Report
| Test | Specifications | Results |
| Description | White to Off White powder | White powder |
| Solubility | Sparingly soluble in water | Complies |
| Identification | IR Spectrum: The infrared sbsorption spectrumof the sample should concordant with that of the spectrum of WS of product | Complies |
| Loss On Drying | NMT 0.5 % w/w | 0.28% w/w |
| Specific Optical Rotation (On Anhydrous Basis) | +10.0 Deg to +12.0 Deg | +10.29 Deg |
| Sulphated Ash | NMT 0.1% | 0.049% w/w |
| Heavy Metals | NMT 20 ppm | < 20 ppm |
| Enantiomeric purity | The area of peak due to R-Pregabalin in NMT 0.5% the area of the S-Pregabalin | 0 |
| Related Substances(By HPLC) | Lactum impurities : NMT 0.5% | Not Detected |
| Individual impurity: NMT 0.5% |
0.298% |
|
| Total Impurities: NMT 1.0% | 0.569% | |
| Assay By HPLC | 98% to 102% w/w | 99.58% w/w |
Looking for "Pregabalin" ?
Explore More Products