Bhiwandi, Thane, Maharashtra
- GST NO. : 27AKFPM8972N1Z9
View Mobile Number
| Business Type | Manufacturer, Exporter, Supplier, Retailer |
From the point of view of marketing, shipping, and handling, our products are securely packed in top-grade supplies using ultramodern packaging machines. The packaging is extremely reliable. Stationed in Mumbai(India), our company is one of the most sought after entity in the industry.
Pharmaceutical Formulations For Ointment & Cream
-
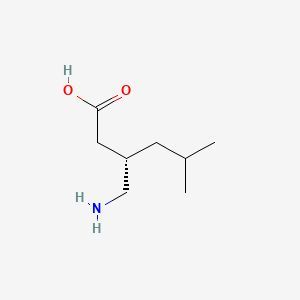
Clotrimazole + Beclomethasone Dipropionate + Gentamicin Sulphate + Chlorocresol (as preservative) Cream
-
Aloe vera + Vitamin E Acetate Moisturising Cream
-
Betamethasone Dipropionate +Salicylic Acid Ointment
-
Clobetasol Propionate + Salicylic Acid Ointment
-
Betamethasone Dipropinate + Gentamicin Cream
-
Betamethasone Dipropinate + Clotrimazole Cream
-
Betamethasone Dipropinate + Gentamicin + Clotrimazole Cream
-
Octyl methoxycinnmate + oxybenzone + avobnzone + vitamin-E aceate + aloe cream
-
Urea + Lactic acid +Propylene Glycol +Light Liquid Paraffin + Preservatives Methyl Paraben +Propyl Paraben Cream
-
Aciclovir Cream B.P. 5% w/w
-
Sliver Sulphadizine and Chlorhexidine Gluconate Cream with Aloe & Allantoin
-
Diclofenac Diethylamine + Oleum Lini + Methyl Salicylate + Menthol+ Capsaicin Gel
-
Povidone-Iodine & Metronidazole Ointment with Aloe
-
Salicylic Acid 12 % Ointment
-
Urea + Lactic Acid + Liquid Paraffin + Propylene Glycol Cream
-
Clindamycin & Nicotinamide in Aloe-Allantoin gel base
-
Clindamycin , Nicotinamide & Aloe SolutionM
-
DiclofenacDiethylamine + Oleum Lini + Methyl Salicylate + Menthol Gel
-
Diclofenac gel B.P
-
Diclofenac Diethylamine + Oleum Lini + Methyl Salicylate + Menthol Gel
-
Salicylic Acid 2 % Gel
Pharmaceutical Formulations For Tablets
| Generic Name | Composition |
| Aceclofenac Tablets 100mg | Each film coated tablet contains : Aceclofenac IP 100mg |
| Albendazole Tablets 400 mg. | Each uncoated chewable tab contains: Albendazole BP 400mg. |
| Amlodipine Tablets 5 mg. | Each uncoated tablet contains: Amlodipine 5mg |
| Artemether &Lumefantrine Tablets | Each uncoated tablet contains : Artemether IP 20mg Lumefantrine 120mg. |
| Atorvastatin Tablets 10 mg | Each film coated tablets contains : Atorvastatin 10mg |
| Ciprofloxacin Tablets BP 500mg | Each film coated tablet contains: Ciprofloxacin Tablets BP. 500mg |
| Indapamide SR Tablets 1.5mg. | Each s/r release film coated tablet contains : Indapamide USP 1.5mg. |
| Levocetirizine Tablets 5mg | Each film coated tablet contains: Levocetrizine Dihydrochloride: 5mg |
| Meloxicam Tablets B.P.7.5 mg |
Each uncoated tablet contains : Meloxicam B.P. 7.5mg |
| Pantoprazole Tablets 20 mg. | Each enteric coated tablets contains Pantoprazole 20mg |
| Paracetamol Tablets BP 650mg | Each Uncoated tablet Contains Paracetamol BP : 650mg |
| Prolonged-Release Isosorbide Mononitrate Tablets BP. |
Each Prolonged Release Tab Contains Diluted Isosorbide Mononitrate BP Eq. to Isosorbide Mononitrate 30mg. |
| Rabeprazole Sodium Tablets 10mg | Each enteric coated tablet contains : Rabeprazole Sodium 10mg |
| Sildinafil Citrate Tablets 100mg | Each Film Coated Tablet Contains: Sildinafil Citrate 100mg. |
| Sodium Valproate Sustained Release Tablets. | Each Enteric Coated tablet Contains Sodium Valproate BP : 333mg Valproic acid USP: 145mg. |
Pharmaceutical Formulations For Injections
| Generic Name | Composition |
| Cefazolin For Injection USP 1gm (Vial + WFI) | Each vial contains : Cefazolin Sodium USP Eq. to Cefazolin 1gm |
| Cefazolin For Injection USP 250mg (Vial + WFI) | Each vial contains : Cefazolin Sodium USP Eq. to Cefazolin 250mg |
| Cefazolin For Injection USP 500mg (Vial + WFI) | Each vial contains : Cefazolin Sodium USP Eq. to Cefazolin 500mg |
| Cefotaxime for Injection USP 1gm (Vial + WFI) | Each vial contains : Cefotaxime Sodium USP Eq. to Cefotaxime 1gm |
| Cefotaxime for Injection USP 250mg (Vial + WFI) | Each vial contains : Cefotaxime Sodium USP Eq. to Cefotaxime 250mg |
| Cefotaxime for Injection USP 500mg (Vial + WFI) | Each vial contains : Cefotaxime Sodium USP Eq. to Cefotaxime 500mg |
| Ceftazidime For Injection USP 1gm (Vial + WFI) | Each vial contains : Ceftazidime Sodium USP Eq. to Ceftazidime 1gmmg (Sterile mixture of Ceftazidime and Sodium Carbonate) |
| Ceftazidime For Injection USP 250mg (Vial + WFI) | Each vial contains : Ceftazidime Sodium USP Eq. to Ceftazidime 250mg (Sterile mixture of Ceftazidime and Sodium Carbonate) |
| Ceftazidime For Injection USP 500mg (Vial + WFI) | Each vial contains : Ceftazidime Sodium USP Eq. to Ceftazidime 500mg (Sterile mixture of Ceftazidime and Sodium Carbonate) |
| Ceftriaxone For Injection USP 1gm Vial + WFI) | Each vial contains : Ceftriaxone Sodium USP Eq. to Ceftriaxone 1gm |
| Ceftriaxone For Injection USP 500mg (Vial + WFI) | Each vial contains : Ceftriaxone Sodium USP Eq. to Ceftriaxone 500mg |
| Cefuroxime For Injection USP 750mg (Vial + WFI) | Each vial contains : Cefuroxime Sodium USP Eq. to Cefuroxime 750mg |
| Pantoprazole Injection 40mg (Vial + WFI) | Each vial contains : Pantoprazole Sodium Sterile 40mg |
| V E T E R I N A R Y | |
| Cefotaxime for Injection USP 5gm | Each vial contains : Cefotaxime Sodium USP Eq. to Cefotaxime 5gm |
| Ceftriaxone For Injection USP 5gm | Each vial contains: Ceftriaxone Sodium USP Eq. to Ceftriaxone 5gm |
| Diminazene Aceturate Injection (For Vet Use Only) | Each ml contains : Diminazene Aceturate B vet C 70mg Phenazone BP 375mg Water for Injection BP q.s. |
Pharmaceutical Formulations For Capsules
| Generic Name | Composition |
| Diltiazem–Hydrochloride Extended Release Capsules USP | Each capsule contains : Diltiazem Hydrochloride BP : 120mg (As Extended Release Pellets) |
| Omeprazole and Domperidone Capsules | Each capsule contains : Omeprazole IP 20mg. (As enteric coated pellets ) Domperidone BP (As pellets ) 10mg. |
| Omeprazole Capsules IP 20 mg. | Each Hard Gelatin Capsule Contains: Omeprazole IP 20mg (An Enteric coated granules) |
| Pantoprazole and Domperidone Capsules | Each hard gelatin capsule contains : Domperidone BP 30mg (As sustained released pellets) Pantoprazole Sodium Sesquihydrate Eq. to Pantoprazole 40mg. (As enteric coated pellets ) |
| Rabeprazole Sodium and Domperidone Capsules | Each hard gelatin capsule contains : Domperidone BP 30mg ( As sustained released pellets ) Rabeprazole Sodium 20mg (As enteric coated pellets ) |
| Salbutamol, Bromohexine and Theophylline Capsules. | Each Hard Gelatin Capsule Contains : Salbutamol Sulphate BP 2.4mg. Bromohexine Hydrochloride BP 8mg. Theophylline BP 100mg. |
| Venlafaxine Extended Release Capsules. 150 mg. | Each Capsules Contains : Venlafaxine Hydrochloride BP equivalent to Venlafaxine: 150mg. ( As Extended Release Pellets) |
| Venlafaxine Extended Release Capsules.75 mg. | Each Capsules Contains : Venlafaxine Hydrochloride BP Equivalent to Venlafaxine 75mg. (As Extended Release Pellets) |
Pharmaceutical Formulations For Syrup
| Generic Name | Composition |
| Antacid Syrup | Each 5ml contains : Dried Aluminum Hydroxide BP 225mg Magnesium Hydroxide BP 200mg Simethicon BP 25mg |
| Bromohexine & Salbutamol Syrup | Each 5ml contains: Bromohexine Hydrochloride BP 4mg. Salbutamol Sulphate BP equivalent to Salbutamol 1mg. |
| Dextromethorphan Hydrobromide, Chlorphenamine Maleate Phenylephrine Hydrochloride and and Menthol Syrup | Each 5 ml Contains : Dextromethorphan Hydrobromide BP 15mg. Chlorphenamine Maleate BP 2mg. Phenylephrine Hydrochloride BP 50mg Menthol BP 2mg |
| Iron ( III) Hydroxide Polymaltose Complex Syrup | Each 5 ml contains : Iron Hydroxide Polymaltose Complex equivalent to Elemental Iron : 50 mg |
| Ofloxacin and Ornidazole Suspension | Each 5 ml contains : Ofloxacin USP 200mg Ornidazole 500mg |
| Salbutamol, Bromohexine and Etophylline Syrup | Each 5 ml Contains : Salbutamol Sulphate BP 1.2mg. Bromohexine Hydrochloride BP 4mg. Etophylline BP 50mg. In mentholated flavored Syrupy base |
| Terbutaline Bromohexine Hydrochloride, Guaiphenesin & Menthol syrup | Each 5 ml Contains : Terbutaline Sulphate BP 1.25mg. Bromohexine Hydrochloride BP 4mg. Guaiphenesin BP 50mg. Menthol BP 2mg |
Looking for "Pharmaceutical Formulations" ?
Explore More Products